.
A bacterial acetyltransferase destroys plant microtubule networks and blocks secretion
Lee AH, Hurley B, Felsensteiner C, Yea C, Ckurshumova W, Bartetzko V, Wang PW, Quach V, Lewis JD, Liu YC, Börnke F, Angers S, Wilde A, Guttman DS, Desveaux D
PLoS Pathog. 2012 Feb;8(2):e1002523
PubMed PMID: 22319451
Abstract
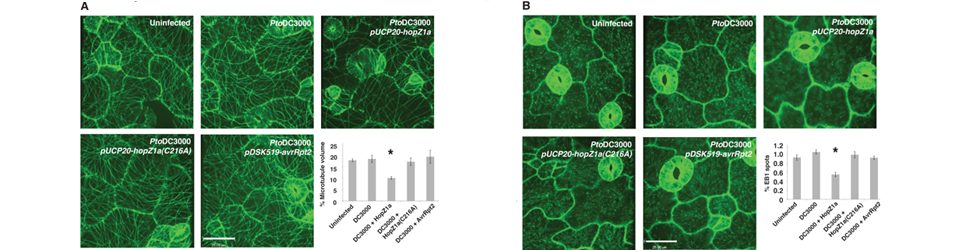
The eukaryotic cytoskeleton is essential for structural support and intracellular transport, and is therefore a common target of animal pathogens. However, no phytopathogenic effector has yet been demonstrated to specifically target the plant cytoskeleton. Here we show that the Pseudomonas syringae type III secreted effector HopZ1a interacts with tubulin and polymerized microtubules. We demonstrate that HopZ1a is an acetyltransferase activated by the eukaryotic co-factor phytic acid. Activated HopZ1a acetylates itself and tubulin. The conserved autoacetylation site of the YopJ / HopZ superfamily, K289, plays a critical role in both the avirulence and virulence function of HopZ1a. Furthermore, HopZ1a requires its acetyltransferase activity to cause a dramatic decrease in Arabidopsis thaliana microtubule networks, disrupt the plant secretory pathway and suppress cell wall-mediated defense. Together, this study supports the hypothesis that HopZ1a promotes virulence through cytoskeletal and secretory disruption.

